What Happens to Neurotransmitters After They Have Been Used
The presynaptic neuron (top) releases a neurotransmitter, which activates receptors on the nearby postsynaptic cell (bottom).
Ligand-gated ion channel showing the binding of transmitter (Tr) and changing of membrane potential (Vm)
Neurotransmission (Latin: transmissio "passage, crossing" from transmittere "send, let through") is the process by which signaling molecules called neurotransmitters are released by the axon concluding of a neuron (the presynaptic neuron), and bind to and react with the receptors on the dendrites of some other neuron (the postsynaptic neuron) a short altitude away. A similar process occurs in retrograde neurotransmission, where the dendrites of the postsynaptic neuron release retrograde neurotransmitters (eastward.g., endocannabinoids; synthesized in response to a rise in intracellular calcium levels) that signal through receptors that are located on the axon terminal of the presynaptic neuron, mainly at GABAergic and glutamatergic synapses.[1] [two] [3] [4]
Neurotransmission is regulated past several dissimilar factors: the availability and rate-of-synthesis of the neurotransmitter, the release of that neurotransmitter, the baseline activeness of the postsynaptic cell, the number of available postsynaptic receptors for the neurotransmitter to bind to, and the subsequent removal or deactivation of the neurotransmitter by enzymes or presynaptic reuptake.[five] [6]
In response to a threshold action potential or graded electric potential, a neurotransmitter is released at the presynaptic terminal. The released neurotransmitter may then motility beyond the synapse to be detected by and bind with receptors in the postsynaptic neuron. Bounden of neurotransmitters may influence the postsynaptic neuron in either an inhibitory or excitatory style. The binding of neurotransmitters to receptors in the postsynaptic neuron can trigger either short term changes, such as changes in the membrane potential called postsynaptic potentials, or longer term changes by the activation of signaling cascades.
Neurons form circuitous biological neural networks through which nervus impulses (action potentials) travel. Neurons do not touch on each other (except in the instance of an electrical synapse through a gap junction); instead, neurons interact at close contact points called synapses. A neuron transports its information by fashion of an action potential. When the nerve impulse arrives at the synapse, it may crusade the release of neurotransmitters, which influence another (postsynaptic) neuron. The postsynaptic neuron may receive inputs from many boosted neurons, both excitatory and inhibitory. The excitatory and inhibitory influences are summed, and if the internet effect is inhibitory, the neuron will be less likely to "fire" (i.eastward., generate an activeness potential), and if the net result is excitatory, the neuron will be more likely to fire. How probable a neuron is to burn down depends on how far its membrane potential is from the threshold potential, the voltage at which an action potential is triggered because enough voltage-dependent sodium channels are activated so that the net inward sodium current exceeds all outward currents.[7] Excitatory inputs bring a neuron closer to threshold, while inhibitory inputs bring the neuron farther from threshold. An action potential is an "all-or-none" event; neurons whose membranes have non reached threshold will not burn, while those that practice must fire. Once the activeness potential is initiated (traditionally at the axon hillock), it will propagate along the axon, leading to release of neurotransmitters at the synaptic bouton to laissez passer along information to nonetheless another adjacent neuron.
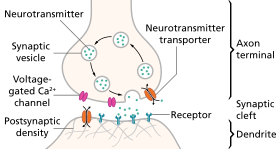
Stages in neurotransmission at the synapse [edit]
- Synthesis of the neurotransmitter. This can have place in the cell torso, in the axon, or in the axon final.
- Storage of the neurotransmitter in storage granules or vesicles in the axon terminal.
- Calcium enters the axon terminal during an activeness potential, causing release of the neurotransmitter into the synaptic cleft.
- Afterward its release, the transmitter binds to and activates a receptor in the postsynaptic membrane.
- Deactivation of the neurotransmitter. The neurotransmitter is either destroyed enzymatically, or taken back into the last from which it came, where it tin can be reused, or degraded and removed.[eight]
General description [edit]
Neurotransmitters are spontaneously packed in vesicles and released in individual quanta-packets independently of presynaptic action potentials. This slow release is detectable and produces micro-inhibitory or micro-excitatory effects on the postsynaptic neuron. An action potential briefly amplifies this procedure. Neurotransmitter containing vesicles cluster effectually agile sites, and afterwards they accept been released may be recycled past one of three proposed mechanisms. The first proposed mechanism involves fractional opening so re-closing of the vesicle. The 2nd two involve the full fusion of the vesicle with the membrane, followed past recycling, or recycling into the endosome. Vesicular fusion is driven largely past the concentration of calcium in micro domains located well-nigh calcium channels, assuasive for only microseconds of neurotransmitter release, while returning to normal calcium concentration takes a couple of hundred of microseconds. The vesicle exocytosis is idea to be driven past a protein complex chosen SNARE, that is the target for botulinum toxins. Once released, a neurotransmitter enters the synapse and encounters receptors. Neurotransmitters receptors tin can either exist ionotropic or g protein coupled. Ionotropic receptors permit for ions to pass through when agonized by a ligand. The main model involves a receptor equanimous of multiple subunits that allow for coordination of ion preference. K poly peptide coupled receptors, also chosen metabotropic receptors, when jump to by a ligand undergo conformational changes yielding in intracellular response. Termination of neurotransmitter activity is usually done past a transporter, however enzymatic deactivation is also plausible.[9]
Summation [edit]
Each neuron connects with numerous other neurons, receiving numerous impulses from them. Summation is the adding together of these impulses at the axon hillock. If the neuron only gets excitatory impulses, it will generate an activeness potential. If instead the neuron gets as many inhibitory as excitatory impulses, the inhibition cancels out the excitation and the nerve impulse will finish there.[10] Action potential generation is proportionate to the probability and design of neurotransmitter release, and to postsynaptic receptor sensitization.[11] [12] [xiii]
Spatial summation means that the effects of impulses received at different places on the neuron add up, so that the neuron may fire when such impulses are received simultaneously, fifty-fifty if each impulse on its own would not be sufficient to cause firing.
Temporal summation means that the effects of impulses received at the same identify can add together up if the impulses are received in close temporal succession. Thus the neuron may fire when multiple impulses are received, even if each impulse on its own would not exist sufficient to cause firing.[14]
Convergence and divergence [edit]
Neurotransmission implies both a convergence and a departure of information. First ane neuron is influenced past many others, resulting in a convergence of input. When the neuron fires, the signal is sent to many other neurons, resulting in a departure of output. Many other neurons are influenced by this neuron.[ commendation needed ]
Cotransmission [edit]
Cotransmission is the release of several types of neurotransmitters from a single nervus terminal.
At the nervus terminal, neurotransmitters are present within 35–l nm membrane-encased vesicles chosen synaptic vesicles. To release neurotransmitters, the synaptic vesicles transiently dock and fuse at the base of specialized 10–fifteen nm cup-shaped lipoprotein structures at the presynaptic membrane called porosomes.[15] The neuronal porosome proteome has been solved, providing the molecular architecture and the complete composition of the machinery.[16]
Contempo studies in a myriad of systems accept shown that most, if not all, neurons release several unlike chemical messengers.[17] Cotransmission allows for more complex furnishings at postsynaptic receptors, and thus allows for more complex advice to occur between neurons.
In mod neuroscience, neurons are often classified past their cotransmitter. For example, striatal "GABAergic neurons" apply opioid peptides or substance P every bit their primary cotransmitter.
Some neurons tin release at least 2 neurotransmitters at the aforementioned time, the other being a cotransmitter, in order to provide the stabilizing negative feedback required for meaningful encoding, in the absence of inhibitory interneurons.[18] Examples include:
- GABA–glycine co-release.
- Dopamine–glutamate co-release.
- Acetylcholine (Ach)–glutamate co-release.
- ACh–vasoactive abdominal peptide (VIP) co-release.
- ACh–calcitonin gene-related peptide (CGRP) co-release.
- Glutamate–dynorphin co-release (in hippocampus).
Noradrenaline and ATP are sympathetic co-transmitters. It is found that the endocannabinoid anadamide and the cannabinoid WIN 55,212-2 tin can modify the overall response to sympathetic nervus stimulation, and indicate that prejunctional CB1 receptors mediate the sympatho-inhibitory action. Thus cannabinoids can inhibit both the noradrenergic and purinergic components of sympathetic neurotransmission.[19]
1 unusual pair of co-transmitters is GABA and glutamate which are released from the aforementioned axon terminals of neurons originating from the ventral tegmental area (VTA), internal globus pallidus, and supramammillary nucleus.[twenty] The quondam two project to the habenula whereas the projections from the supramammillary nucleus are known to target the dentate gyrus of the hippocampus.[20]
Genetic association [edit]
Neurotransmission is genetically associated with other characteristics or features. For example, enrichment analyses of dissimilar signaling pathways led to the discovery of a genetic association with intracranial book.[21]
Meet likewise [edit]
- Autoreceptor
- Biological neuron model § Synaptic transmission
- Electrophysiology
- G poly peptide-coupled receptor
- Molecular neuropharmacology
- Neuromuscular manual
- Neuropsychopharmacology
References [edit]
- ^ Melis M, Pistis M (Dec 2007). "Endocannabinoid signaling in midbrain dopamine neurons: more than physiology?". Current Neuropharmacology. five (four): 268–77. doi:x.2174/157015907782793612. PMC2644494. PMID 19305743.
Thus, it is conceivable that depression levels of CB1 receptors are located on glutamatergic and GABAergic terminals impinging on DA neurons [127, 214], where they can fine-tune the release of inhibitory and excitatory neurotransmitter and regulate DA neuron firing.
Consistently, in vitro electrophysiological experiments from contained laboratories accept provided evidence of CB1 receptor localization on glutamatergic and GABAergic axon terminals in the VTA and SNc. - ^ Flores A, Maldonado R, Berrendero F (Dec 2013). "Cannabinoid-hypocretin cross-talk in the central nervous arrangement: what we know so far". Frontiers in Neuroscience. 7: 256. doi:x.3389/fnins.2013.00256. PMC3868890. PMID 24391536.
Direct CB1-HcrtR1 interaction was beginning proposed in 2003 (Hilairet et al., 2003). Indeed, a 100-fold increase in the potency of hypocretin-1 to activate the ERK signaling was observed when CB1 and HcrtR1 were co-expressed ... In this study, a higher authorisation of hypocretin-i to regulate CB1-HcrtR1 heteromer compared with the HcrtR1-HcrtR1 homomer was reported (Ward et al., 2011b). These data provide unambiguous identification of CB1-HcrtR1 heteromerization, which has a substantial functional impact. ... The existence of a cantankerous-talk betwixt the hypocretinergic and endocannabinoid systems is strongly supported by their partially overlapping anatomical distribution and mutual role in several physiological and pathological processes. Nevertheless, little is known virtually the mechanisms underlying this interaction. ... Interim every bit a retrograde messenger, endocannabinoids attune the glutamatergic excitatory and GABAergic inhibitory synaptic inputs into the dopaminergic neurons of the VTA and the glutamate manual in the NAc. Thus, the activation of CB1 receptors present on axon terminals of GABAergic neurons in the VTA inhibits GABA transmission, removing this inhibitory input on dopaminergic neurons (Riegel and Lupica, 2004). Glutamate synaptic transmission in the VTA and NAc, mainly from neurons of the PFC, is similarly modulated by the activation of CB1 receptors (Melis et al., 2004).
• Figure 1: Schematic of brain CB1 expression and orexinergic neurons expressing OX1 (HcrtR1) or OX2 (HcrtR2)
• Figure two: Synaptic signaling mechanisms in cannabinoid and orexin systems
• Figure 3: Schematic of brain pathways involved in food intake - ^ Freund TF, Katona I, Piomelli D (July 2003). "Part of endogenous cannabinoids in synaptic signaling". Physiological Reviews. 83 (3): 1017–66. doi:x.1152/physrev.00004.2003. PMID 12843414.
- ^ Ayakannu, Thangesweran; Taylor, Anthony H.; Marczylo, Timothy H.; Willets, Jonathon Yard.; Konje, Justin C. (2013). "The Endocannabinoid Organization and Sex Steroid Hormone-Dependent Cancers". International Journal of Endocrinology. 2013: 259676. doi:10.1155/2013/259676. ISSN 1687-8337. PMC3863507. PMID 24369462.
- ^ Nagatsu, T. (December 2000). "[Molecular mechanisms of neurotransmission]". Rinsho Shinkeigaku = Clinical Neurology. 40 (12): 1185–1188. ISSN 0009-918X. PMID 11464453.
- ^ Andreae, Laura C.; Burrone, Juan (March 2018). "The role of spontaneous neurotransmission in synapse and excursion development". Journal of Neuroscience Research. 96 (three): 354–359. doi:10.1002/jnr.24154. ISSN 0360-4012. PMC5813191. PMID 29034487.
- ^ Holden A, Winlow W (1984). The Neurobiology of Pain: Symposium of the Northern Neurobiology Group Held at Leeds on 18 April 1983 (1st ed.). Manchester Univ Pr. p. 111. ISBN978-0719010613.
- ^ Kolb B, Whishaw IQ (2003). Fundamentals of Homo Neuropsychology (5th ed.). Worth. pp. 102–104. ISBN978-0-7167-5300-1. (reference for all five stages)
- ^ Squire L, Berg D, Bloom FE, du Lac S, Ghosh A, Spitzer NC (2013). Key neuroscience (quaternary ed.). Amsterdam: Elsevier/Academic Press. pp. 133–181. ISBN978-0-12-385870-2.
- ^ Williams SM, McNamara JO, Lamantia A, Katz LC, Fitzpatrick D, Augustine GJ, Purves D (2001). Purves D, Augustine GJ, Fitzpatrick D, et al. (eds.). Summation of Synaptic Potentials. Neuroscience (2nd ed.). Sunderland (MA): Sinauer Associates.
- ^ Wang JH, Wei J, Chen Ten, Yu J, Chen Due north, Shi J (September 2008). "Gain and fidelity of transmission patterns at cortical excitatory unitary synapses amend fasten encoding". Periodical of Cell Scientific discipline. 121 (Pt 17): 2951–60. doi:10.1242/jcs.025684. PMID 18697836.
- ^ Yu J, Qian H, Chen N, Wang JH (2011). "Quantal glutamate release is essential for reliable neuronal encodings in cerebral networks". PLOS ONE. 6 (9): e25219. Bibcode:2011PLoSO...625219Y. doi:10.1371/journal.pone.0025219. PMC3176814. PMID 21949885.
- ^ Yu J, Qian H, Wang JH (August 2012). "Upregulation of transmitter release probability improves a conversion of synaptic counterpart signals into neuronal digital spikes". Molecular Encephalon. 5 (26): 26. doi:x.1186/1756-6606-5-26. PMC3497613. PMID 22852823.
- ^ Hevern VW. "PSY 340 Brain and Behavior". Archived from the original on Feb nineteen, 2006.
- ^ Anderson LL (2006). "Discovery of the 'porosome'; the universal secretory machinery in cells". Journal of Cellular and Molecular Medicine. 10 (one): 126–31. doi:x.1111/j.1582-4934.2006.tb00294.x. PMC3933105. PMID 16563225.
- ^ Lee JS, Jeremic A, Shin L, Cho WJ, Chen X, Jena BP (July 2012). "Neuronal porosome proteome: Molecular dynamics and architecture". Journal of Proteomics. 75 (xiii): 3952–62. doi:10.1016/j.jprot.2012.05.017. PMC4580231. PMID 22659300.
- ^ Trudeau LE, Gutiérrez R (June 2007). "On cotransmission & neurotransmitter phenotype plasticity". Molecular Interventions. 7 (3): 138–46. doi:10.1124/mi.vii.iii.5. PMID 17609520.
- ^ Thomas EA, Bornstein JC (2003). "Inhibitory cotransmission or later-hyperpolarizing potentials tin can regulate firing in recurrent networks with excitatory metabotropic transmission". Neuroscience. 120 (2): 333–51. doi:ten.1016/S0306-4522(03)00039-3. PMID 12890506. S2CID 26851745.
- ^ Pakdeechote P, Dunn WR, Ralevic V (November 2007). "Cannabinoids inhibit noradrenergic and purinergic sympathetic cotransmission in the rat isolated mesenteric arterial bed". British Journal of Pharmacology. 152 (5): 725–33. doi:x.1038/sj.bjp.0707397. PMC2190027. PMID 17641668.
- ^ a b Dh, Root; South, Zhang; Dj, Barker; J, Miranda-Barrientos; B, Liu; Hl, Wang; M, Morales (2018-06-19). "Selective Brain Distribution and Distinctive Synaptic Architecture of Dual Glutamatergic-GABAergic Neurons". Cell Reports. 23 (12): 3465–3479. doi:10.1016/j.celrep.2018.05.063. PMC7534802. PMID 29924991.
- ^ Adams HH, Hibar DP, Chouraki V, Stein JL, Nyquist PA, Rentería ME, et al. (December 2016). "Novel genetic loci underlying homo intracranial volume identified through genome-wide clan". Nature Neuroscience. 19 (12): 1569–1582. doi:10.1038/nn.4398. PMC5227112. PMID 27694991.
External links [edit]
- Historical evolution of the neurotransmission concept
Source: https://en.wikipedia.org/wiki/Neurotransmission
0 Response to "What Happens to Neurotransmitters After They Have Been Used"
Postar um comentário